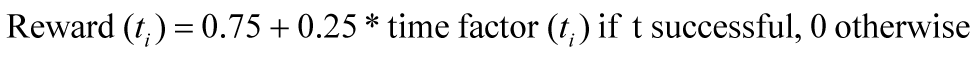| Table of Contents |  |
|
Original Article
| ||||||
| Goal-oriented feedback on motor behavior in virtual reality based stroke therapy: A case study using the rehabilitation gaming system | ||||||
| Sebastian Marwecki1, Belén Rubio Ballester1, Esther Duarte2, Paul F.M.J. Verschure1,3 | ||||||
|
1Laboratory of Synthetic Perceptive, Emotive and Cognitive Systems, Center of Autonomous Systems and Neurorobotics, Pompeu Fabra, Roc Boronat, Barcelona, Spain
2Servei de Medicina Física I Rehabilitació, Hospitals del Mar I l´Esperança, Institut Hospital del Mar d´Investigacions Mèdiques, Barcelona, Spain 3ICREA, Institucio´ Catalana de Recerca i Estudis Avanc¸ats, Passeig Llui´s Companys, Barcelona, Spain | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Marwecki S, Ballester BR, Duarte E, Verschure PFMJ. Goal-oriented feedback on motor behavior in virtual reality based stroke therapy: A case study using the rehabilitation gaming system. Edorium J Disabil Rehabil 2017;3:36–45. |
|
ABSTRACT
|
|
Aims:
We address the role of short-term goals in virtual reality (VR) applications for motor relearning, which benefit stroke therapy.
| |
|
Keywords:
Augmented feedback, Goal theory, Meaningful play, Motor relearning, Stroke rehabilitation, Virtual reality
| |
|
INTRODUCTION
| ||||||
|
Current trends in stroke incidence suggest that by 2030 there will be 70 million stroke survivors worldwide [1]. Stroke survivors suffer from deficits which include, but are not limited to, aphasia, memory impairments as well as loss of strength and motor functions due to hemiparesis. Patients regain motor control in supervised rehabilitation, which, at later stages, can be relocated to ambulatory settings. Especially in those later stages, virtual reality (VR) based systems have been reported to improve the process of relearning lost motor functions, as they can provide massed and individualized practice to the patients in a safe and ecologically valid environment [2]. Furthermore, skills learned in VR are transferable to similar real world tasks and even generalizable to other real world tasks [3]. A number of studies even have indicated VR based therapy to be more effective than conventional therapy[4] [5]. Given that VR-based systems for stroke therapy are to be used in ambulatory settings, research has depicted essential affordances those systems need to fulfill [6] . To enhance patients´ intrinsic motivation (including: perceived effort, the apparent amount of energy invested in the task, and perceived competence) [7] , mentioned systems should provide “relevant rehabilitation goals„[8] as done by therapists in conventional therapy [6]. Understandable short-term goals render feedback “meaningful„ [9] as they depict therapy progress, which fosters adherence [8][9][10]. However, apart from these goals being seldom implemented in VR based systems for stroke therapy, research has not yet focused on which different types of short-term goals may provide the most benefit. Nicholls et al. defined two major types of short-term goals: mastery and ego goals [11]. The objective in mastery goals is to increase one´s ability, hence learning, while ego goals serve as a meaning of skill demonstration. The former supports self-comparison with regard to previous performances, while in ego goals one´s performance is compared to those of others. Whether in research or commercially available, a multitude of applications using VR based systems for stroke therapy exist [2]. The rehabilitation gaming system (RGS) fosters upper-limb stroke recovery by providing motor relearning tasks in a variety of settings [12]. For example, users are incentivized to hit virtual spheres by awarding them with points. Many similar systems require those reaching movements towards virtual objects, which, if successful, are rewarded by a scoring system (Table 1). Overall, the output of the implemented scoring systems does not relate to a short-term goal, which could be considered meaningful to the patient. Successful tasks (additional points) do not relate to one´s past performance or the performance of other patients, hence, no mastery or ego goal is provided. Contrarily, an application in the SMART system [9] provides this comparison by summarizing the user´s performance and comparing it to an optimal one. This comparison can now be done concurrently (while playing) within automated VR-based scenarios, thus providing meaningful feedback and goals, facilitating a context for establishing mastery and ego goals. In mastery goals, one tries to increase perceived competence by investing more effort. This increase of intrinsic motivation then may lead to massed practice [7], which has been shown to facilitate learning [13] and retention of motor gains [14]. Contrarily, ego goals may constitute avoidance of challenge [15] due to unfavorable social comparison and therefore a decrease in learning. This negative affect is more prevalent, when one has low ability [16], which arguably holds true for stroke patients. One should however consider that VR can be adapted to the user´s needs, thereby providing favorable comparisons in ego goals. This work therefore aims at answering the following research questions:
| ||||||
|
MATERIALS AND METHODS
| ||||||
|
Healthy participants | ||||||
|
A study with healthy participants was carried out to test the hypotheses in a controlled setting. Twelve healthy participants (9 females, 27.4±8.7 years) participated in this study. Their eyesight was normal or corrected to normal and no cognitive or speech deficits were apparent. This information was self-reported. All healthy participants showed a clear preference in handedness (1 left-handed), which was assessed using the Edinburgh handedness inventory [17], which showed absolute values above 50 (clear preference) for all participants (9 females, M = 0.76, SD = 0.30). Further inclusion criteria were: no apparent cognitive or speech deficits that would interfere with the understanding of the task. | ||||||
|
Stroke patients | ||||||
|
After conducting the study with healthy participants, the study was adapted to fit the needs of stroke patients. Eight chronic stroke patients with hemiparesis (2 left hemiparetic, 1 female, maximum age of 80 years) from the rehabilitation department at the Hospital de l´Esperança in Barcelona participated in the study. All patients were non-aphasic and had no apparent cognitive or visual deficits. Two participants did not fully complete the experimental session due to exhaustion, so that one participant only completed one block of trials for the control condition and another only one block for both the mastery and control condition. Data from both participants are included in the results, as neither showed outlier characteristics. The ethics committee of clinical research of the Parc de Salut Mar approved experimental guidelines. | ||||||
|
Setup | ||||||
|
For the healthy participants, movement data was tracked using an imager (Microsoft Kinect), while audiovisual feedback was provided using headphones and a head-mounted display (HMD), the Oculus Rift DK2 (Oculus VR, Invine, CA). The HMD is used to occlude vision of the real arm movements. For stroke patients, a chair mounted arm support (Armeo Spring, Hocoma AG, Zurich, CH) was used to track the movement of the patients more precisely. Audiovisual feedback was provided using speakers and a 2D display, as patients showed signs of discomfort with the HMD. Differences in the setup configuration for patients and healthy participants did not allow for direct comparison between groups (Figure 1). | ||||||
|
Task description | ||||||
|
To provide the means to compare accumulated rewards to a reference value, the experimental setup utilized an adapted version of a training scenario of the RGS [18], a playful application comprising time-critical unimanual planar reaching tasks towards virtual targets (Figure 2). Patients were instructed to use their paretic arm. Healthy participants were instructed to use their non-dominant, as the non-dominant upper-limb is generally less precise than the dominant one [19] and we wanted to avoid ceiling effects in performance and have more control over the manipulation of the movement precision. Vertical movements of the virtual limb were omitted. The virtual targets appeared within a fixed distance from the starting point, which was adapted to the user, and within a range of 130°. After each trial, the user had to return his hand to the start position. For deriving the speed of motor adaptation, a 30° horizontal perturbation was added to the mapping of the real and the virtual limb. Each play-through, or block, comprised 30 trials. Before the first and three other randomly set trials of each block, the direction of the perturbation changed. Instructions were given before and summary feedback was given after each block. With regard to the participant, the application provided instruction and feedback in either English, Spanish or Catalan. Audiovisual feedback was provided on reaching or missing the virtual target. An overlaid visualization depicted the progress towards a mastery or ego goal (Figure 2). Designed to have a low cognitive affordance, this visualization consisted of a simple slider next to a picture indicating whether the user was performing better or worse in comparison to their own baseline performance. While for the mastery goal the user´s picture was shown, for the ego goal another person´s picture was shown to let the user believe he or she is competing against that other person. In this study, this person was someone who already participated and provided the consent to use his or her picture. However, or both mastery and ego goal, this comparison was always based on the user´s own baseline performance. | ||||||
|
Reward computation | ||||||
|
After each successful trial, a positive reward was given, which depends on the time used for the reaching task, while an unsuccessful trial leads to no reward. This reward was then compared to the average of the performance baseline, to derive the acute score of the user. where ta = total amount of trials  Before displaying it, the score was then normalized with regard to the total possible positive or negative score and enhanced by an exponential function, to make changes in the acute score easier to distinguish.    | ||||||
|
Procedure | ||||||
|
The participants were introduced to the experiment and their written consent was obtained. Each participant started with a short training period of 18 trials. A baseline condition of one block then tested their performance to provide reference for the mastery and ego condition. Three conditions were then tested: A control condition, in which no goal is provided, a “mastery„ condition, in which reference to a former performance is provided, and an “ego„ condition, in which reference to the performance of another person is provided. All three conditions were tested in a within-subjects design with repeated measures using three repetitions of three blocks for healthy participants, and two repetitions of two blocks for stroke patients (Figure 3). Due to time constraints, for stroke patients the ego condition was excluded and the performance baseline served as a repetition of the control condition. After the participant underwent all trials, he or she was debriefed. Participants did not receive financial or other material compensation for their participation. The ethics committee of clinical research of the Parc de Salut Mar and Vall d´Hebron Research Institute approved experimental guidelines. | ||||||
|
Measurements | ||||||
|
According to the aforementioned hypotheses, measurements include perceived effort, perceived competence, four different kinematic variables and motor learning. | ||||||
|
Perceived effort and perceived competence | ||||||
|
Subscales of the IMI [7] are used to determine perceived effort (5-item subscale) and perceived competence (6-item subscale). Participants were provided with both subscales after the final third iteration of each condition block. A translation of these questionnaires to Spanish was provided if needed. Please notice that the ego condition was only tested in healthy participants and perceived effort and perceived competence were only measured in healthy participants. These changes in the experimental design were made to avoid physical fatigue in patients and to reduce the duration of the experimental session. | ||||||
|
Kinematic variables | ||||||
|
To further investigate on whether the different conditions influence motor behavior, four different variables are computed to express task performance: reaction time, response time, movement extent and accuracy (Figure 4).
The reaction time is defined by the time passed between the start of the trial (pstart) and the moment after which the participant starts moving his hand forward, the initialization of movement (pinit). | ||||||
|
Motor learning | ||||||
|
The measurement of motor learning utilizes the concept of adaptation to random visual perturbations of the trajectory in a reaching task. | ||||||
|
Data analysiss | ||||||
|
For the analysis, all distributions were checked for normality using a Shapiro–Wilk test (a = 0.05). Consecutively, repeated measures analyses of variance (ANOVA) and Friedman tests were carried out. The latter was namely applied for analysis of perceived effort of healthy participants and two kinematic variables in stroke patients. Since the inclusion criteria and demographics were different between groups of participants (i.e., stroke patients and healthy participants) we never performed statistical comparisons between groups. All other dependent variables were normally distributed. Post-hoc analysis entails one sided and two-sided dependent t-tests or Wilcoxon signed rank tests without z-scores due to exact computation for small sample sizes. One sided tests include comparisons between mastery to control and ego to control condition for both intrinsic motivation subscales and all kinetic variables. All other comparisons were two-sided. Statistical thresholds for p-values were adjusted using Bonferroni correction. A one-way ANOVA was performed for the measurement of potential order effects. | ||||||
|
RESULTS
| ||||||
|
To test the feasibility of using short-term mastery or ego goals in VR stroke therapy, we analyzed their effect on perceived effort and perceived competence, kinematic variables and motor learning. No order effects were apparent. Results are given in (Table 2). | ||||||
|
Perceived effort and perceived competence (healthy participants only) | ||||||
|
For perceived effort, post-hoc analysis between conditions revealed differences between mastery condition and control condition (p = 0.007), and ego and control condition (p = 0.008), with both the mastery and ego condition achieving higher mean scores than the control. Interestingly, no difference between mastery and ego condition were apparent (p = 1.00). For perceived competence, comparing ego and control condition showed a trend (p = 0.018) with the ego condition performing slightly better, whereas no improvement from the mastery to the control condition (p = 0.194) or difference between mastery and ego condition (p = 0.255) could be derived from the data. | ||||||
|
Kinematic variables | ||||||
|
Analyzing differences between conditions yielded a significant difference in reaction time (p = 0.027) and a trend in response time (p =0.063) for healthy participants. Post-hoc analysis showed that the mastery condition outperformed the control condition in both reaction time (p = 0.007) and response time (p = 0.014). Furthermore, performance in the ego condition was better than the control condition in response time (p = 0.023), where it showed a trend, but not in reaction time (p = 0.056). Also, performance in the mastery condition was not different than in the ego condition for either reaction time (p = 0.16) or response time (p = 0.465). No significant differences were obtained for accuracy and movement extent. For stroke patients, no kinematic variable showed significant differences between conditions. Neither in accuracy (p = 0.945), reaction time (p = 0.578), response time (p = 0.673) or movement extent (p = 0.186), was the average performance in the mastery condition better, or worse, than in the control condition. Since participants stated that their attention towards the visual feedback decreased as they became accustomed to it, the computations for the kinematic variables and motor learning were again carried out with regard only to the first iteration of each condition. Contrary to before, stroke patients showed better average performance in response time when comparing the mastery to the control condition (t (7) = -2.93, p = 0.011, M = -0.15, SD = 0.15, not depicted in Table 2). Other variables yielded no noteworthy difference. | ||||||
|
Motor learning | ||||||
|
Based on the obtained data, no difference between conditions could be found for either healthy participants (p = 0.323) or stroke patients (p = 0.316). Post-hoc analysis of the data of healthy participants also did not reveal differences between any two conditions, neither between mastery and control (p = 0.554), ego and control (p = 0.101), or mastery and ego (p = 0.342). While both patients and healthy participants similarly seem to adapt stronger to a high error magnitude in the former trial, they do not show different adaptation between conditions (Figure 5). | ||||||
|
| ||||||
|
| ||||||
| ||||||
| ||||||
|
| ||||||
|
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
Based on the above stated results, personalized mastery goals may increase perceived effort as well as kinetic variables connected to task performance, as assumed in our first hypothesis. While data from healthy participants was more conclusive, due to higher sample size, also stroke patients showed increased task performance in further analysis. These findings strongly emphasize considering self-referencing mastery goals in the design process of VR based stroke therapy, as they may provide additional incentives to adhere to the given tasks. Furthermore, we observed no difference in task performance or intrinsic motivation between mastery and ego goals. These results suggest that ego goals may have a positive impact in VR based motor learning applications, as assumed in our second hypothesis; however further studies should confirm this results in stroke patients undergoing motor rehabilitation. Challenge establishes the intensity of training, which in turn determines recovery. This study controlled the level of challenge, ergo the effectiveness of the system as a therapy tool. Accordingly, participants did not seem to alter the motor learning behavior. These findings raise the question, if not challenge adaptation alone is the main factor contributing to the effectiveness of VR based stroke therapy. Recent studies, which looked into the effectiveness of VR based stroke therapy systems, indirectly altered the level of challenge, e.g. by changing the systems interface [20] or by introducing joint game play [21][22]. By using mediators of challenge, which can be manifold and comprise the interface or the modality of feedback, one automatically confounds measurement of the therapy tools effectiveness. Going further, if motivators such as goal-setting have only little direct influence on the therapy tools effectiveness, research should restrain from analyzing motivators which do not directly affect challenge. Systems can already make use of various heuristics and guidelines for motivational game and application design. We should, therefore, strive for more simplicity in the research for the design of therapy applications and games. Academia already suggests, that not a single game, but constant change of therapy games, and therefore modality and nature of feedback and reward, should be used to foster adherence [23]. In follow-up studies, shortcomings of the study design should be addressed. The partial randomization of the study design for stroke patients affected condition order as the study always started with the control condition. This might have influenced the obtained results. As the application was used in a supervised setting, biases regarding intrinsic motivation might be existent. Measuring motivation in ambulatory settings without supervision, also by using the IMI for stroke patients, may provide more applicable results. Effort optimization exploiting electromyography data of adjacent muscle regions, while not feasible to derive actual metabolic cost [24], could improve measurement of motor learning [25]. Goal-setting has been proven to affect self-efficacy [26], which in turn may positively affect compliance [27] and quality of life of stroke patients [28]. Analyzing this influence in VR based stroke therapy will therefore also be beneficial. | ||||||
|
CONCLUSION
| ||||||
|
We have shown that virtual reality (VR) based stroke therapy for ambulatory use benefit from the use of personalized mastery as well as ego goals as they may add to its long-term effectiveness. Furthermore, we argued for future analysis to focus on intrinsic reinforcement, which directly mediate challenge. This may also benefit patients with other various needs to which VR based therapy is applicable. | ||||||
|
REFERENCES
|
|
|
[HTML Abstract]
[PDF Full Text]
|
| Acknowledgements |
| We would like to express their gratitude to all participants who took part in the study, especially the patients and their therapists at the rehabilitation center at the Hospital de l´Esperança in Barcelona. |
|
Author Contributions
Sebastian Marwecki – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Belén Rubio Ballester – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Esther Duarte – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Paul F.M.J. Verschure – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest |
|
Copyright
© 2017 Sebastian Marwecki et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|